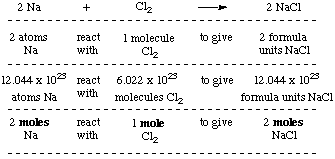SEM images of the samples electro-deoxidized in the CaCl 2-NaCl-1mol%... | Download Scientific Diagram

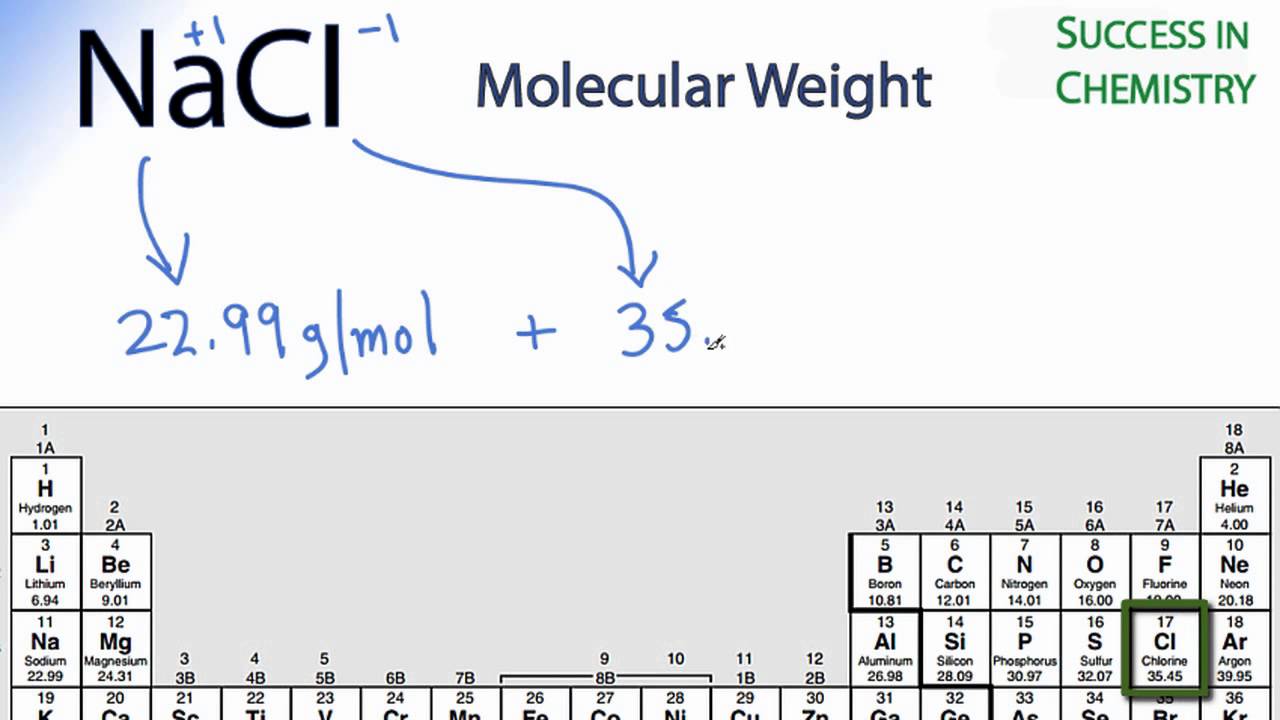
MOLE CALCULATIONS. Moles to Mass Find the mass of one mole of NaCl 58.5g Find the mass of two moles of NaCl 117.0g How did you get the answer? 2 moles. -

Static CA experiments at 1 mol/L NaCl and 45°C at muscovite surface.... | Download Scientific Diagram
If the relative decrease in vapour pressure is 0.4 for a solution containing 1mol NaCl in 3 mol H2O,

Explain why on addition of 1 mol of NaCl to 1 litre of water, the boiling point of water increases, while addition of 1 mol of methyl alcohol to one litre of

How will the boiling point of a liter of water containing 1 mole of sodium chloride (NaCl) compare with that of a liter of water containing 1 mole of calcium chloride (CaCl2)?

Explain in why on addition of 1 moe of NaCl to 1L of water, the boiling point of water increases, - YouTube