Phase contrast microscopy of Caco-2 cells at different passage numbers.... | Download Scientific Diagram

Growing season Δ¹⁴C of ambient CO2 in Pasadena, CA, a city within the... | Download Scientific Diagram

Ca(OH)2+CO2=CaCO3+H2O balance the equation by algebraic method or a,b,c method.ca(oh)2+co2=caco3+h2o - YouTube
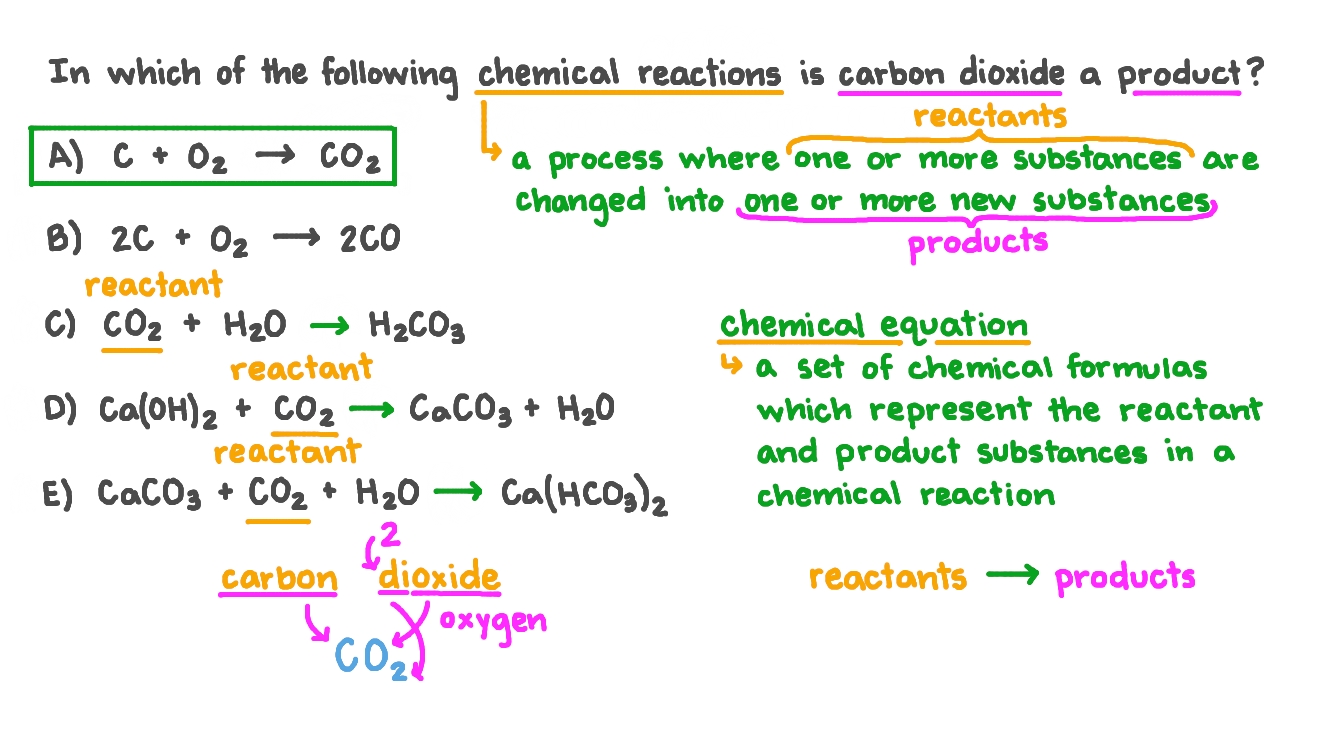
Nutrients | Free Full-Text | Modulatory Effects of Osthole on Lipopolysaccharides-Induced Inflammation in Caco-2 Cell Monolayer and Co-Cultures with THP-1 and THP-1-Derived Macrophages

SOLVED: Calculate ΔHrxn for Ca(s) + ½ O2(g) + CO2(g) → CaCO3(s) given the following set of reactions: Ca(s) + ½ O2(g) → CaO(s) ΔHrxn = −635.1 kJ CaCO3(s) → CaO(s) +
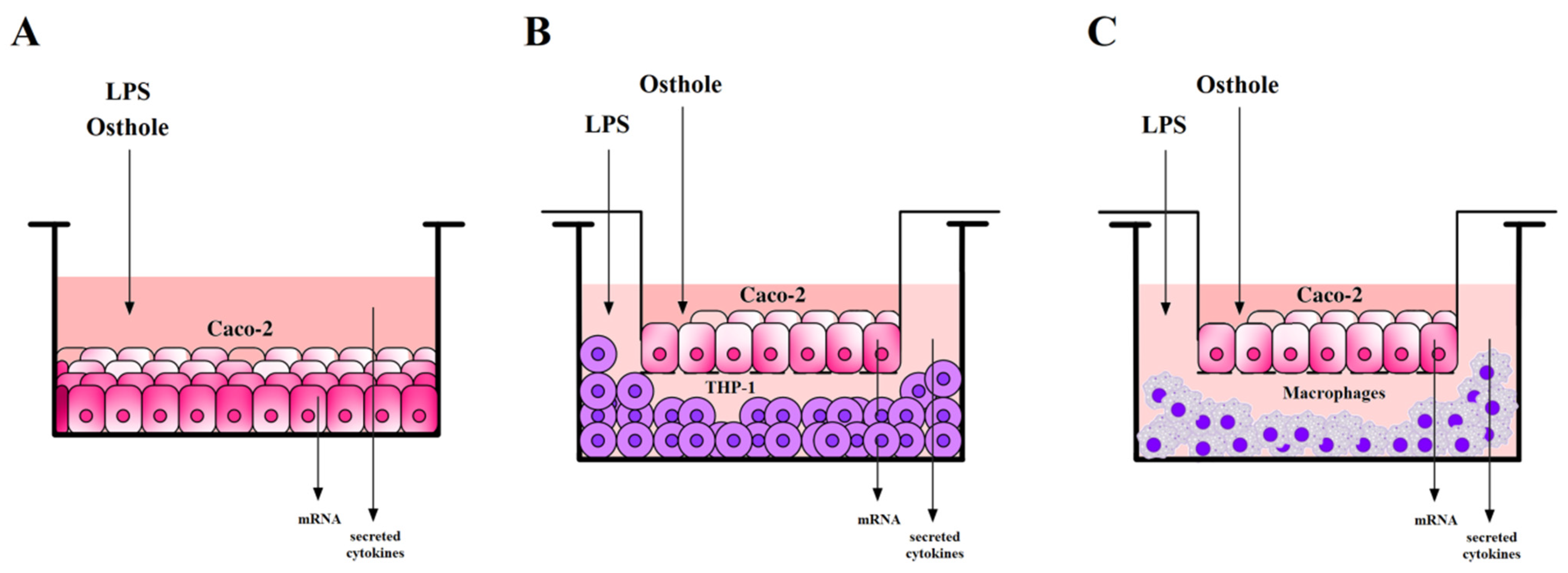
An air CO2 capture system based on the passive carbonation of large Ca(OH)2 structures - Sustainable Energy & Fuels (RSC Publishing)

Carbonation Rates of Dry Ca(OH)2 Mortars for CO2 Capture Applications at Ambient Temperatures | Industrial & Engineering Chemistry Research

![CELL SEARCH SYSTEM -CELL BANK- (RIKEN BRC) [RCB0988 : CACO-2] CELL SEARCH SYSTEM -CELL BANK- (RIKEN BRC) [RCB0988 : CACO-2]](https://cellbank.brc.riken.jp/cell_bank/Upload/Images/gazo/RCB0988_1.jpg)

![Caco-2 [Caco2] - HTB-37 | ATCC Caco-2 [Caco2] - HTB-37 | ATCC](https://www.atcc.org/-/media/product-assets/images/micrographs/cell-biology/htb-37-low-high.jpg?rev=ec2ca58333d54e44abea7f379cbac5ee)