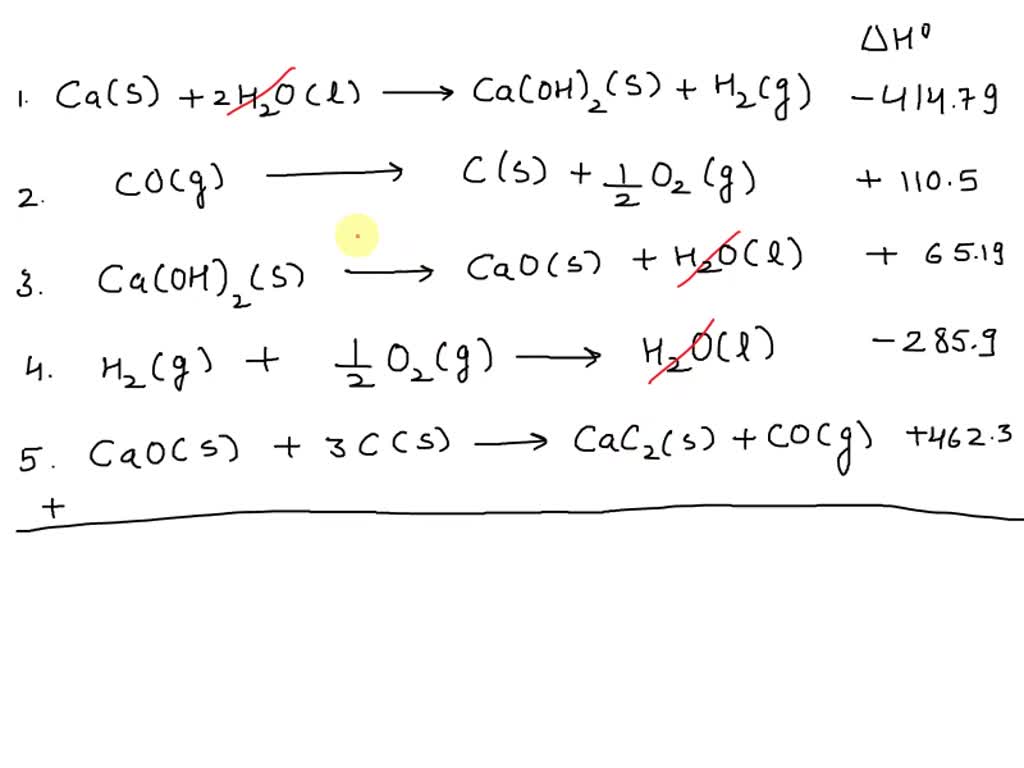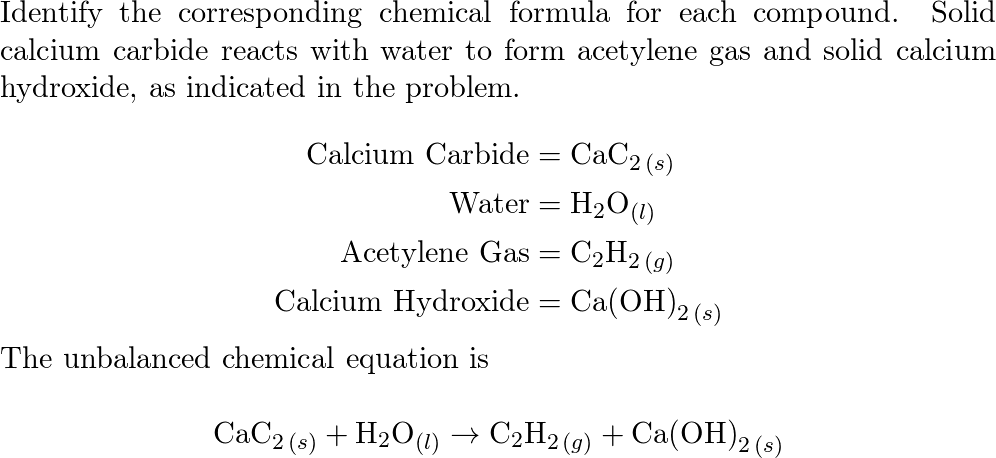Towards C1 chemistry: methanol vinylation by CaC2 in water in the presence of potassium or sodium carbonates - Parshina - 2019 - Journal of Chemical Technology & Biotechnology - Wiley Online Library
Complete the following reaction and name the products A, B and C. Cac2 + ( H2O) → A + (hot Cu tube) → B + ((conc. H2SO4 + HNO3)/(323 - 333K)) → C - Sarthaks eConnect | Largest Online Education Community
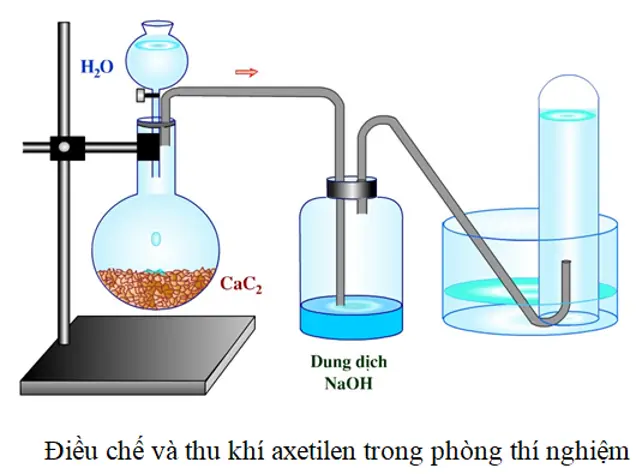
Phản ứng CaC2 + H2O → C2H2 + Ca(OH)2? Cách tạo khí axetylen từ phản ứng CaC2 + H2O - Cao đẳng Nghề Việt Mỹ

Fluoride-Assisted Activation of Calcium Carbide: A Simple Method for the Ethynylation of Aldehydes and Ketones | Organic Letters

Class11Cac2+H2O=acetylene (calcium carbide react with water)complete reaction explanations in Telugu - YouTube
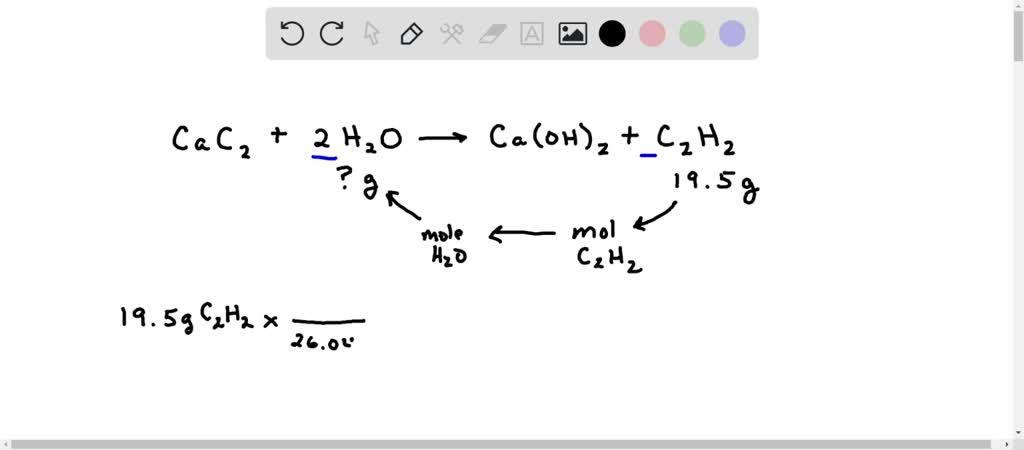
SOLVED: Calcium carbide (CaC2) reacts with water to form acetylene (C2H2): CaC2 (s) + 2 H2O (g) → Ca(OH)2 (s) + C2H2 (g) How many grams of water are required to produce
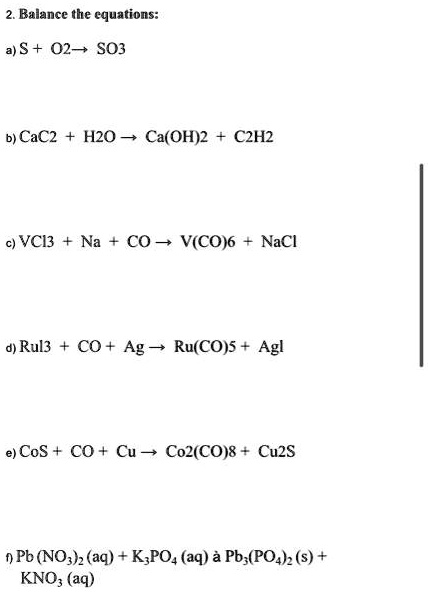
SOLVED: Balance the equations: a) S + 02- S03 b) CaC2 H2O Ca(OH)2 C2H2 VCI3 Na + CO V(CO)6 NaCl Rul3 CO + Ag Ru(CO)s Agl CoS CO + Cu Coz(CO)8 Cuzs
CaC2 reacts with H2O and gives X. X reacts with Cu2Cl2 in the presence of NH4OH and give Y. What is Y, and how? - Quora

Bell ringer # 2 : How many grams of CaC2 are needed to produce 78.0 g of acetylene? CaC2(s) + 2 H2O(l) → C2H2(g) + Ca(OH)2(aq) TIME'S UP! START TIMER ppt download