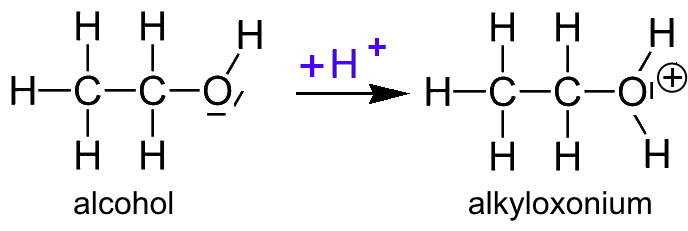HOMCOM Portable Tabletop Ventless Bio Ethanol Fireplace with Glass Walls Stainless Steel Arc Base Bronze | Oriental Trading

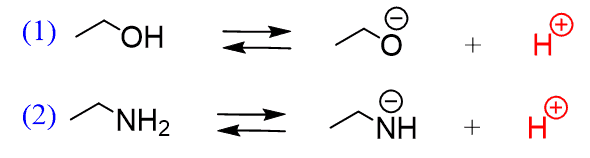
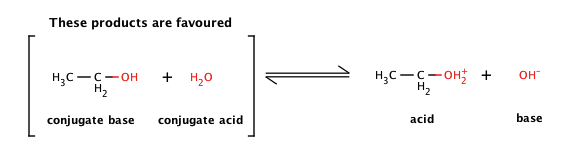
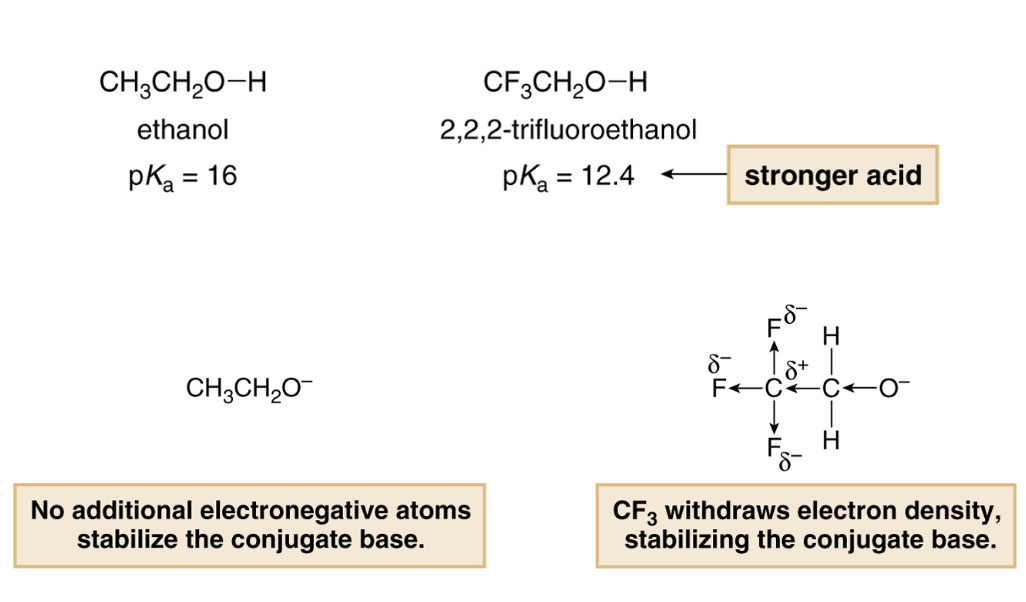
1,1,1-Trichloroethanol is an acid more than 1000 times stronger than ethanol, even though both have a conjugate base where the negative charge is on an oxygen. Provide an explanation for this observation.

Mechanism of reaction between caprolactam and ethanol in the presence... | Download Scientific Diagram
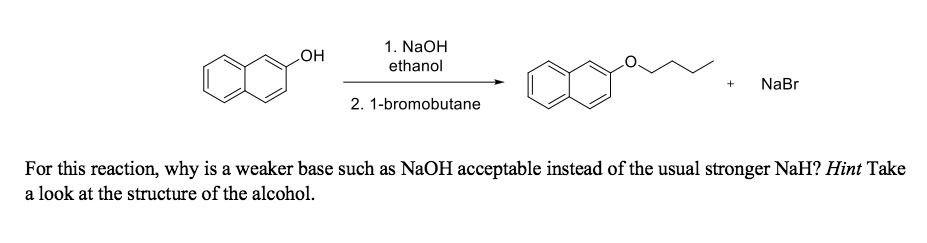
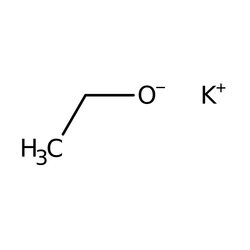
SOLVED: NaOH ethanol OH NaBr 2. 1-bromobutane For this reaction; why is a weaker base such as NaOH acceptable instead of the usual stronger NaH? Hint Take look at the structure of