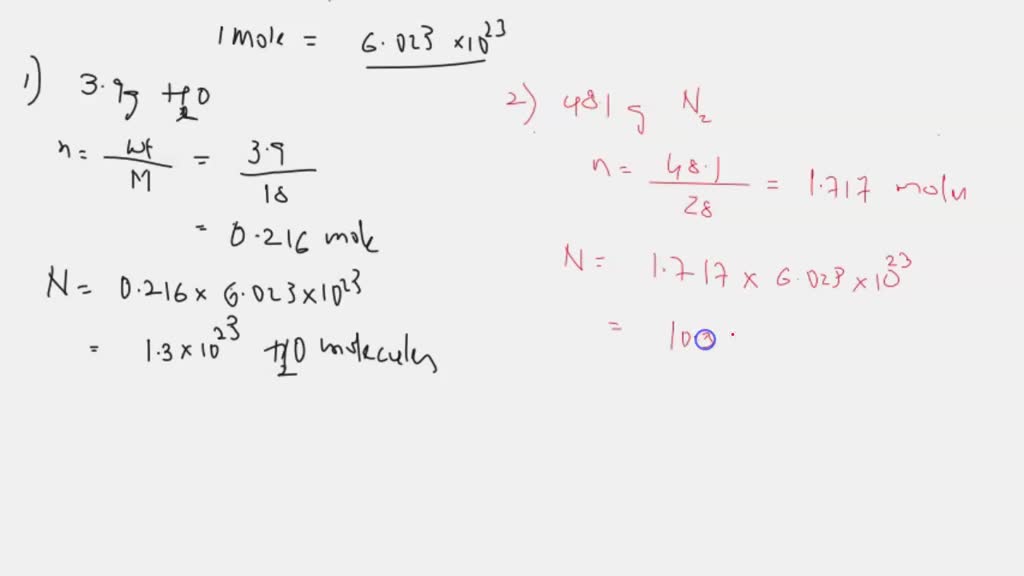
SOLVED: How many molecules are in each sample? A) 3.9g H2O Express using two significant figures. N= molecules B) 48.1g N2 Express using three significant figures. N= molecules C) 89g CCl4 Express

SOLVED: When 17.07 g Al2S3 reacts with 15.54 g H2O, all the Al2S3 is used up, how many grams of excess H2O remain at the end of the reaction? Al2S3 + 6H2O —–>
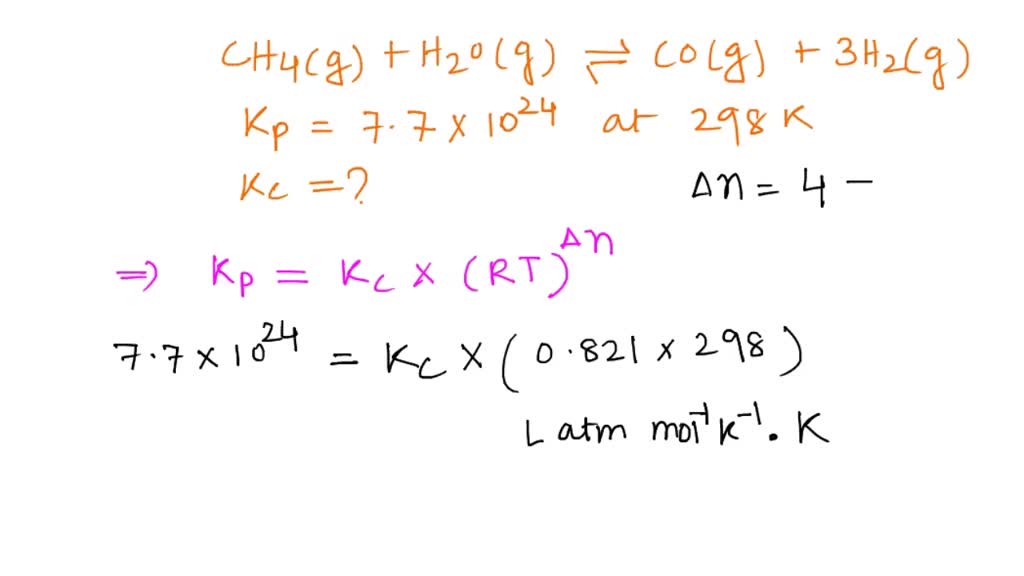
SOLVED: Consider the reaction below. H2O (g) + CH4 (g) <—> CO (g) + 3H2 (g) Kc = 4.7 at 1400 K What is Kp for this reaction at 1400 K? 6.2 x 104 4.7 8.2 x 10^8

See: Calculate the amount of heat released when 27.0 g H2O is cooled from a liquid at 314 K to a solid at - Brainly.com
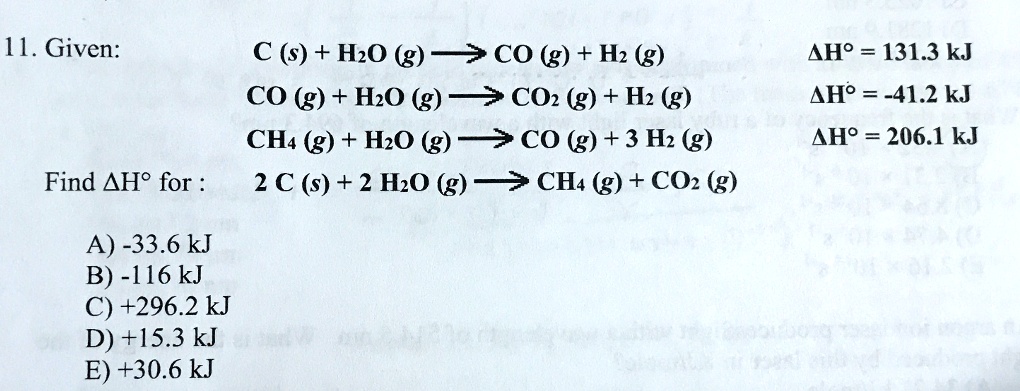
SOLVED: 11. Given: C (s) + H2O (g) ->CO (g) + Hz (g) CO (g) + HzO (g) > COz (g) + Hz (g) CHa (g) + HzO (g) >C0 (g) +