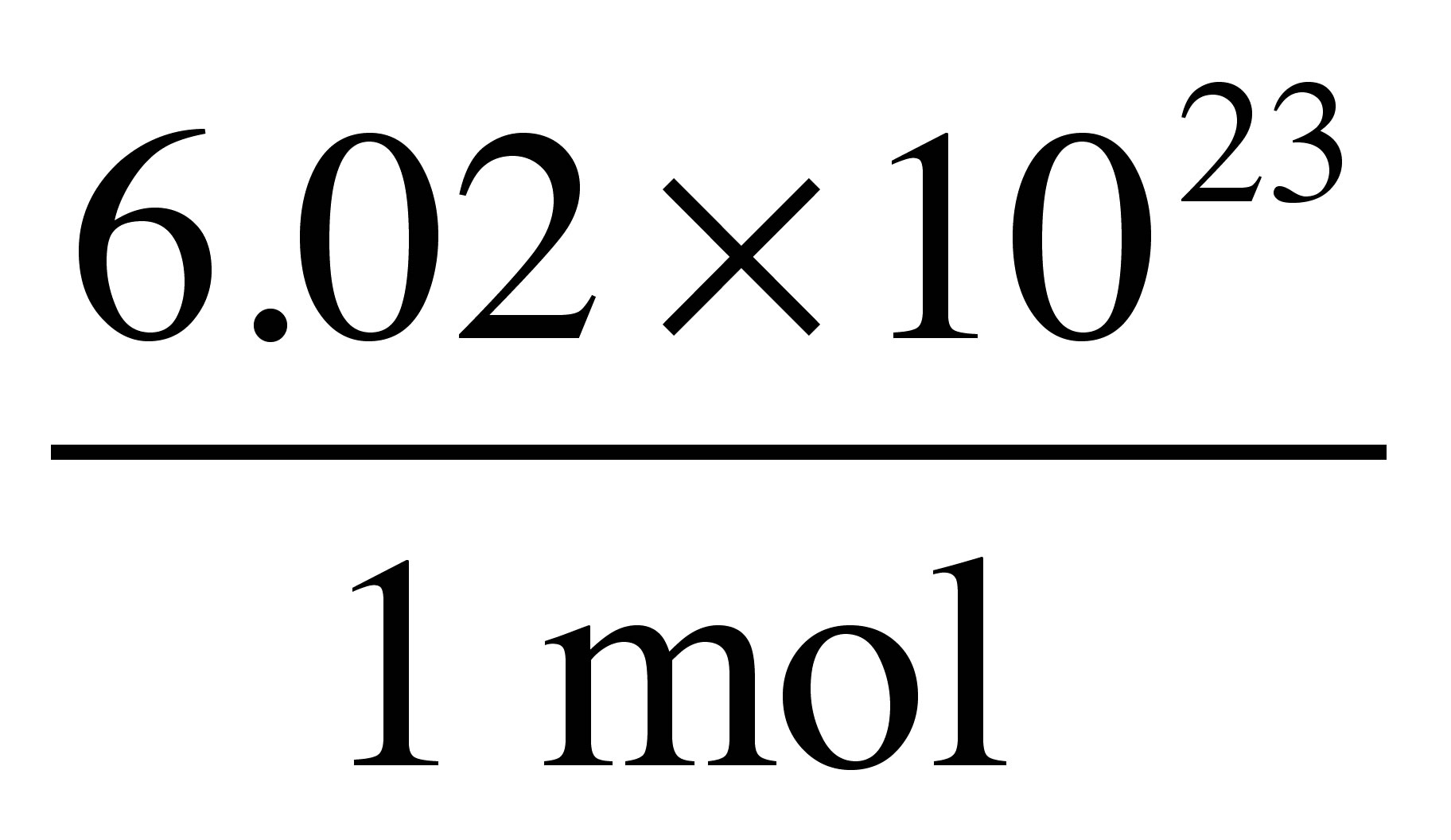Wet air containing 4.0 mole% water vapor is passed through a column of calcium chloride pellets. The pellets absorb 97.0% of the water and none of the other constituents of the air.

Question Video: Determining the Mass of Water Lost When a Hydrated Compound of Cobalt(II) Sulfate Is Heated | Nagwa

Question Video: Calculating the Mass of Water Produced Given the Masses of Oxygen and Hydrogen | Nagwa